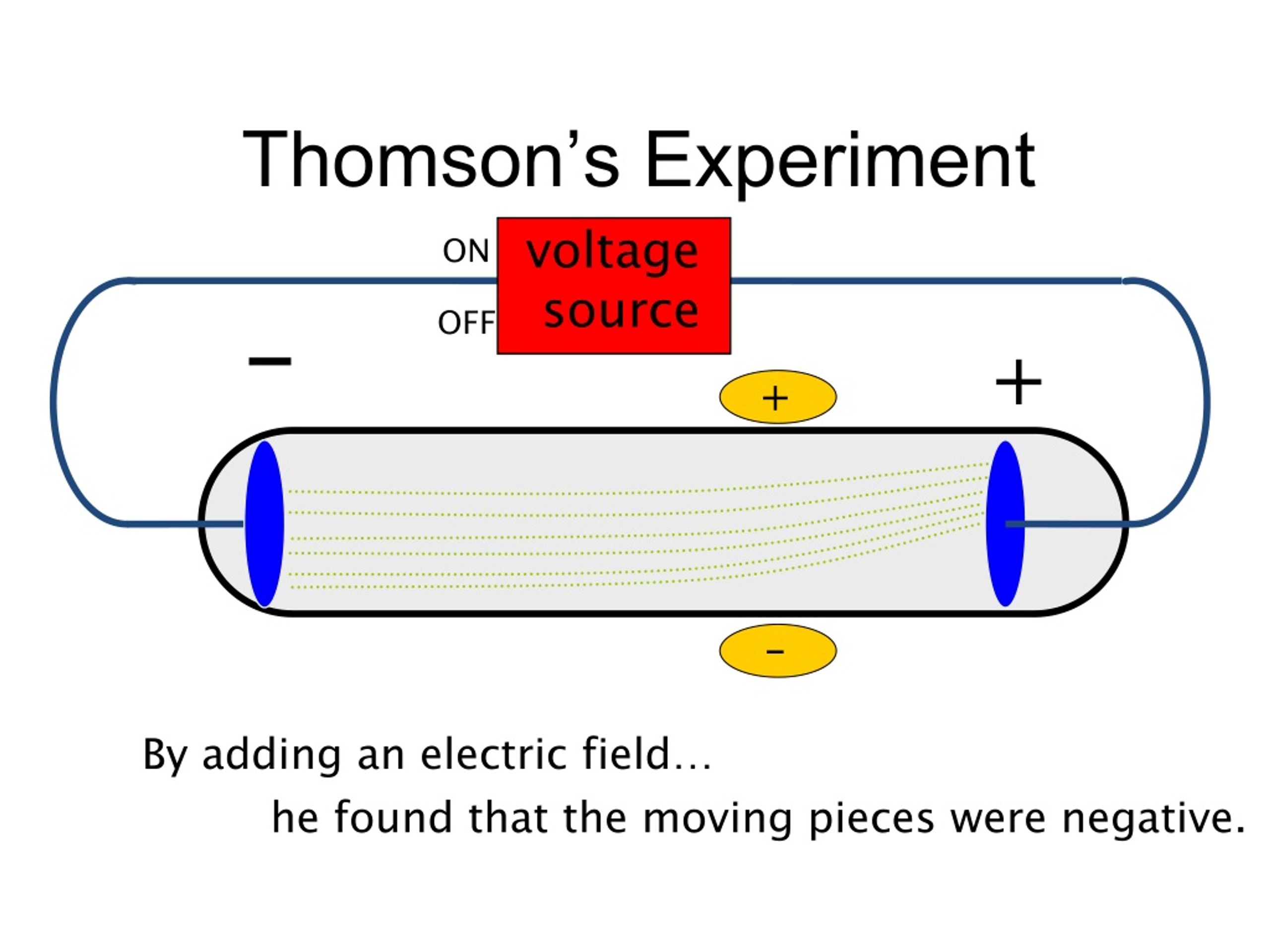
Irst, in a variation of an 1895 experiment by Jean Perrin, Thomson built a cathode ray tube ending in a pair of metal cylinders with a slit in them. He advanced the idea that cathode rays are really streams of very small pieces of atoms. Thomson reason that electrons must be a part of the atoms of all elements? Charge-to-mass ratio of electrons was the same, regardless of the gas used.O atoms have parts? J.J. Why did JJ Thomson reason that electrons? What was Thompson working with when he discovered the cathode rays? Thompson was working with gas discharge tubes. What was Thompson working with when he discovered the cathode rays? The atomic theory states that all material substances are made of atoms and that each element has a unique atomic structure. Thomson’s cathode ray tube experiments provided the first evidence that atoms were composed of even smaller particles called electrons. What did JJ Thomson’s cathode ray tube experiment show quizlet? He also determined that the charge to mass ratio of electrons which was much greater (about 2000 times) than that of proton. ✨ Answer ✨ Thomson’s experiment revealed that cathode rays are composed of negatively charged particles called electrons. Why did JJ Thomson experiment with cathode ray tubes Brainly? What evidence from the Cathode Ray Tube Experiment support the claim that electrons have a negative charge? The Cathode Ray bent way from a negatively charged plate. What evidence is found in the cathode ray tube? (ii) At anode: Anode decreases in size due to the formation of copper ions. See also There Money In Quantum Physics What is the observation at the cathode?Īt cathode: Greyish white metal lead is formed on the cathode. one was negatively charged, the other was positively charged, and he noticed that these particles that left the cathode ended up bending toward the positive electrode. He had to have electrodes That were orthogonal or 90° to the other electrodes. How did JJ Thomson know the electron was negative? Is cathode ray positive or negative?Ĭathode rays are negatively charged particles because they are attracted to the plate of the cathode ray tube. He called his atom the plum pudding model. To explain the neutrality of atoms, Thomson proposed a model of the atom in which negative electrons are scattered throughout a sphere of positive charge. Thomson discovered the first subatomic particle, the electron, while researching cathode rays. This approach can be applied to solve the question. As per the cathode ray experiment the electrons are negatively charged for a cathode ray. Hint: In this question the gases hydrogen and helium are filled in the tubes, one needs to find out whether this will have any effect on the cathode ray experiment. 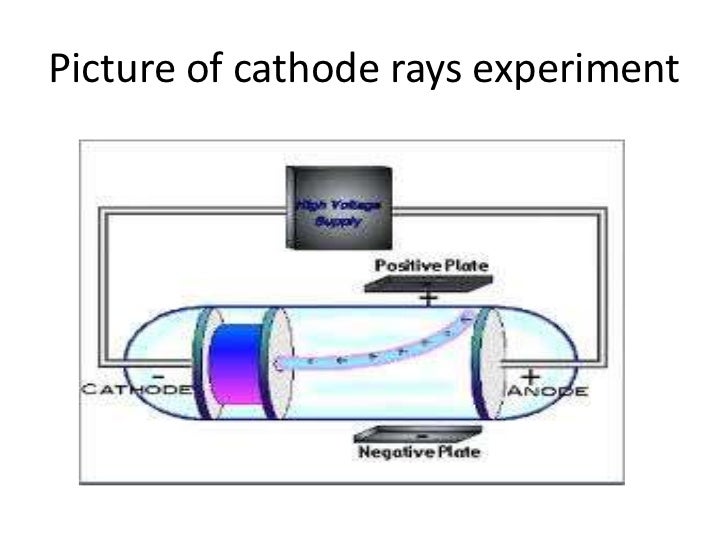
Thomson’s second experiment demonstrated that the cathode rays bent away from a negatively charged metal plate and towards a positively charged plate which meant cathode rays were negatively charged. What happened in the cathode ray tube experiment? Using this information Thomson determined the mass to charge ratio of an electron. Then Thomson measured how much various strengths of magnetic fields bent the particles. Thomson was able to deflect the cathode ray towards a positively charged plate deduce that the particles in the beam were negatively charged. See also Is Google Earth app free? How did Thomson discover electrons with cathode ray? In 1897 he reported that “cathode rays” were actually negatively charged particles in motion he argued that the charged particles weighed much less than the lightest atom and were in fact constituents of atoms. What did JJ Thomson conclude about cathode rays? This proved that the negative charge and the ray were inseparable and intertwined. Thomson’s First Cathode Ray Experiment He found that by applying a magnetic field across the tube, there was no activity recorded by the electrometers and so the charge had been bent away by the magnet. He found that when the rays entered the slit in the cylinders, the electrometer measured a large amount of negative charge. Thomson wanted to see if, by bending the rays with a magnet, he could separate the charge from the rays. Perrin had found that cathode rays deposited an electric charge. Why did JJ Thomson experiment with cathode ray tubes? 
Why did JJ Thomson reason that electrons?.What was Thompson working with when he discovered the cathode rays?.What did JJ Thomson’s cathode ray tube experiment show quizlet?.Why did JJ Thomson experiment with cathode ray tubes Brainly?.What evidence is found in the cathode ray tube?.What is the observation at the cathode?.How did JJ Thomson know the electron was negative?.What happened in the cathode ray tube experiment?.How did Thomson discover electrons with cathode ray?.What did JJ Thomson conclude about cathode rays?.What experiment did JJ Thomson perform?.Why did JJ Thomson experiment with cathode ray tubes?.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
